Haloakanes and haloarenes are halogen containing organic compounds used in industries, medicine, in agriculture. Chloamphenicol is a chlorine containing antibiotic which is used in the treatment of typhoid fever.
- Chloroquine is used for the treatment of malaria
- Halothane is used as anesthetic during surgery.
- Thyroxine is iodine containing hormone whose deficiency cause goiter
- DDT and BHC are used as Insecticides.
Contents
Haloalkanes and Haloarenes Test-1

Topic:- IUPAC Nomenclature
Write the IUPAC Name of following compounds:
Q.1 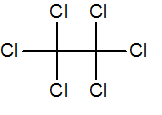
Show Answer
hexachloroethane
Q.2 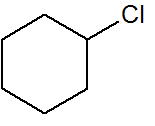
Show answer
chlorocyclohexane
Q.3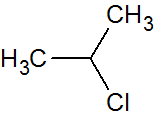
Show answer
2-chloropropane
Q.4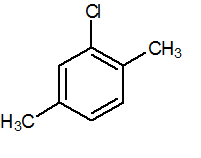
Show answer
2-chloro-1,4-dimethylbenzene
Q.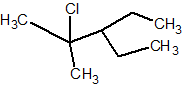
Show answer
2-chloro-3-ethyl-2-methylpentane
Q.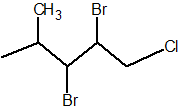
Show answer
2,3-dibromo-1-chloro-4-methylpentane
Q.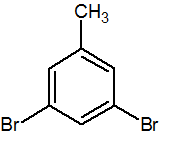
Show answer
1,3-dibromo-5-methylbenzene
Q.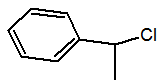
Show answer
(1-chloroethyl)benzene or 1-chloro-1-phenyl ethane
Q.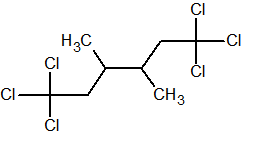
Show answer
1,1,1,6,6,6-hexachloro-3,4-dimethylhexane
Haloalkanes and Haloarenes Test-2

Topic:- Reasoning Questions
Explain giving reasons:- (each question carries 2 marks)
Q.1 Thionyl chloride is preferred for converting alcohol to haloalkane
Ans.1 Thionyl chloride is preferred for converting alcohol to haloalkane because the biproducts SO2 is gas which escape into the atmosphere. HCl is absorbed by pyridine.
R –OH + SOCl2 ---> RCl +SO2+HCl
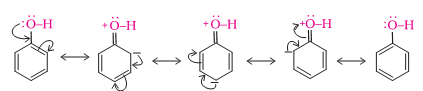
Q.2 Phenol cannot be converted to chlorobenzene by reacting with HCl.
In phenol, due to resonance, the carbon –oxygen bond has a partial double bond character and is difficult to break. Therefore it can not be converted to chlorobenzene by reacting with HCl.
Q.3 HNO3 is added during iodination of benzene.
When benzene is reacted with iodine, the reaction is reversible in nature. Therefore and oxidizing agent like HNO3 oxidizes the
HI formed in the reaction and keeps the reaction in forward direction.
Q.4 p- dichlorobenzene has higher melting point than meta – dichlorobenzene
p- dichlorebenzene is having symmetrical structure therefore it can fit better into the crystal lattice which increases its melting point.
Q.5 The boiling points of isomeric haloalkenes decrease with increase in branching.
The boiling points of isomeric haloakanes decreases with branching due to decrease in surface areas with branching .As branching increasing the structure becomes more spherical and the surface area decreases. e.g. the boiling points of isomers of C4H9 Br follows the order.
Q.6 Hydrolysis of optically active 2- bromobutane forms optically inactive butan – 2 – ol.
The compound 2- bromobutane undergoes hydrolysis by SN1 mechanism via the formation of carbocation which is planar.

The attack of nucleophile can result in product which is a mixture of compounds both with same configuration and inverted configuration.
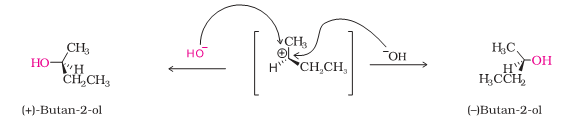
Therefore it results in the formation of racemic mixture which is optically inactive.
Q.7 Chlorobenzene is less reactive towards nucleophilic substitution reaction.
Chlorobenzene is less reactive towards nucleophillic substitution due to
a) Due to Resonance , C- Cl bond acquires a double bond character
b) Unstability of phenyl cation
c) More s-character
Q.8 Chloroform is stored in dark coloured bottles.
Chloroform gets oxidsed by air in the presence of light and form poisonous gas phosgene. Therefore to protect from sunlight, it is kept in dark coloured bottles.
Q.9 The order of boiling points is RCl < RBr < RI.
Q.10 Vinyl chloride is less reactive than allyl chloride




